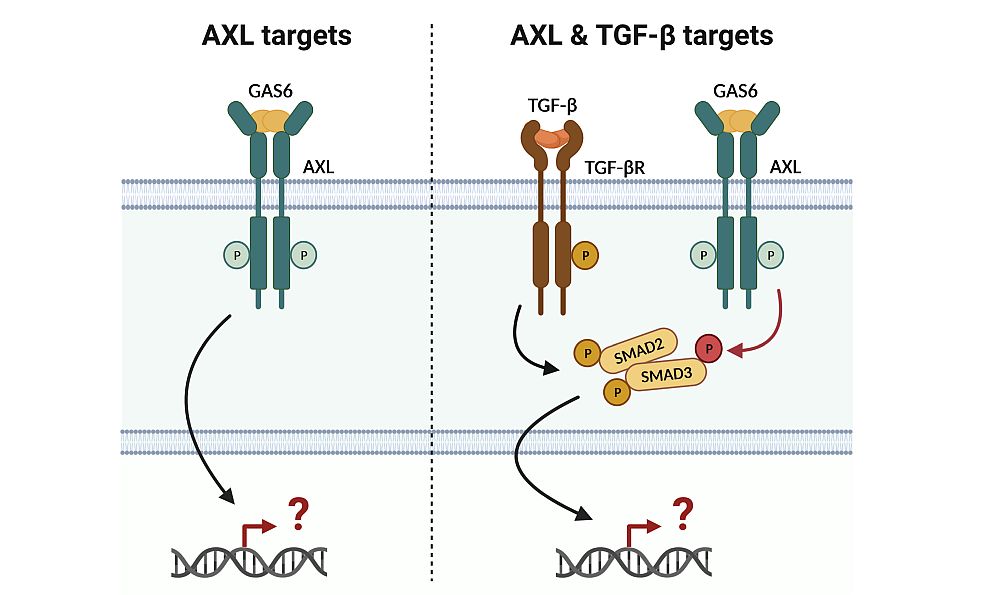
Question & Aim: Identify Target Genes of Gas6/AXL and TGF-β Signaling in Hepatocellular Carcinoma
A large proportion of patients with hepatocellular carcinoma (HCC) show upregulation and activation of Gas6/AXL which can molecularly cooperate with TGF-β signaling by the aberrant phosporylation of the Smad3 linker region. Both AXL and TGF-β signaling have multifaceted functions in cancer cells and in the tumor microenvironment by modulating tumor invasion and immune response. We focus on the tumor intrinsic role of AXL and AXL/TGF-β signaling to identify target genes in cancer cells which allow the development of pinpointed therapeutic strategies without overt cellular side effects.
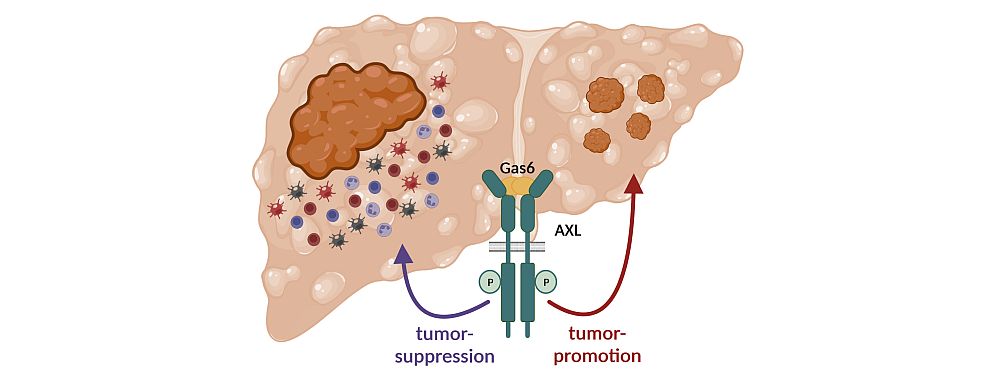
Question & Aim: The Role of AXL in the Development of Hepatocellular Carcinoma
AXL is a major driver of epithelial to mesenchymal transition (EMT) of tumor cells which is a crucial event in cancer invasion and metastasis. In immune cells, Gas6/AXL expression is tightly linked to the phagocytosis of apoptotic cells by macrophages and is a key inducer of the anti-inflammatory response. In HCC, the role of Gas6/AXL signaling in EMT and in the immune microenvironment remains to be investigated. We aim to gain a profound understanding of the role of tumor cell-specific AXL as well as the role of AXL on stromal cells during hepatic fibrosis progression and liver tumorigenesis. Understanding the mechanisms of Gas6/AXL signaling in liver pathophysiology allow us to evaluate whether AXL serves as a valuable target to treat HCC patients.
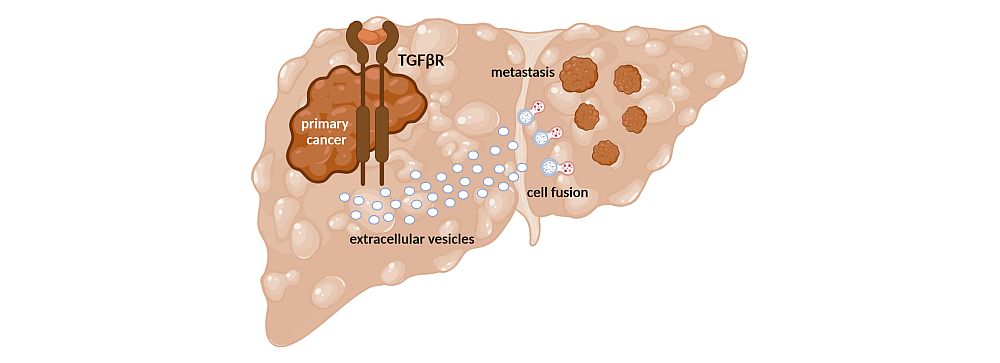
Question & Aim: TGF-β in Extracellular Vesicle-Mediated Cancer Dissemination of Hepatocellular Carcinoma
Extracellular vesicles (EVs) are central in TGF-β-mediated hepatocellular carcinoma (HCC) dissemination. Tumor cell-derived EVs are crucially involved in long range signaling by fusion or interaction with recipient cells in order to establish a pre-metastatic niche (PMN). The understanding of the EV cargo – regulated by TGF-β – in the PMN formation at distal sites is of outmost relevance for the understanding HCC dissemination. We study the long range signals of HCC cells which are sent through EVs to establish a PMN-like tumor environment that is essentially involved in hepatic colonization of metastatic cancer cells.
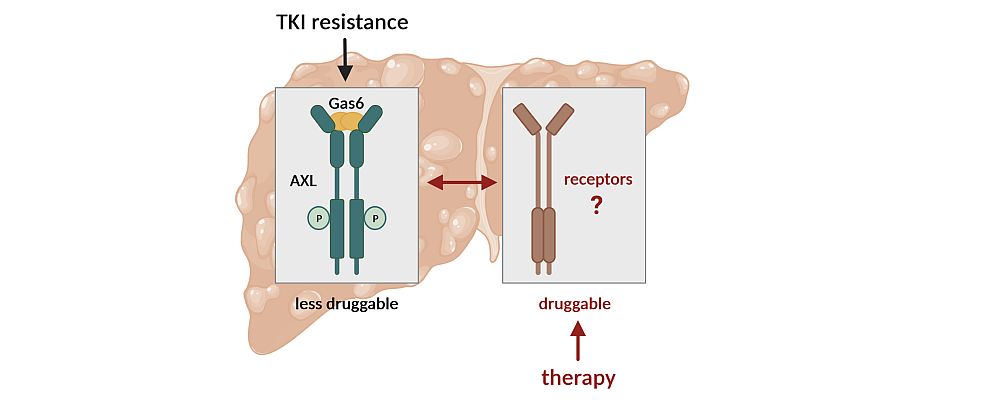
Question & Aim: Facing Gas6/AXL Signaling in the Drug Resistance of Hepatocellular Carcinoma
Patients with hepatocellular carcinoma (HCC) at advanced stage require systemic therapy by treatment with the targeted tyrosine kinase inhibitors (TKIs) Sorafenib or Lenvatinib at first-line and Regorafenib or Cabozantinib at second-line. Although systemic TKI treatments have clinical benefits, the improvement in patient outcomes is modest due to limited efficacies. The major cause of treatment failure in systemic therapy settings is drug resistance resulting from acquired mechanisms. The causes of the high refractoriness of HCC to novel targeted TKIs based on the use of Lenvatinib, Regorafenib and Cabozantinib are poorly understood. We aim to identify the vulnerabilities linked to Gas6/AXL in the escape from TKI treatments in order to develop novel strategies to efficiently cope with HCC.
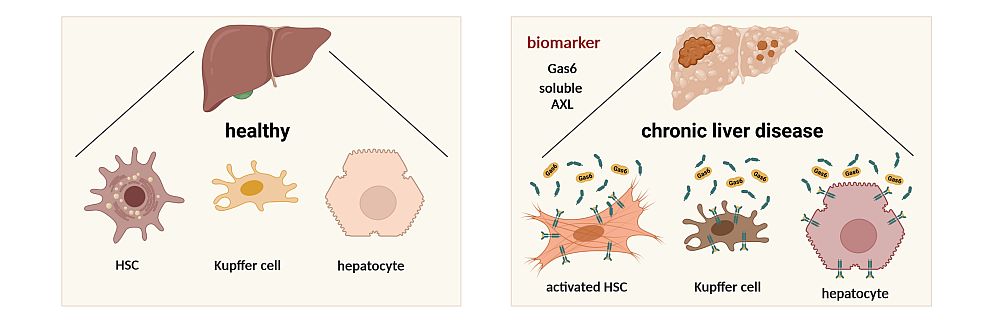
Question & Aim: Soluble AXL and Gas6 as Biomarkers of Chronic Liver Disease and Hepatocellular Carcinoma
The expression of AXL is post-translationally regulated by shedding of the extracellular domain resulting in the release of soluble AXL (sAXL). Enhanced levels of sAXL in combination with the ligand of the AXL receptor – known as Gas6 – have been detected in liver cirrhosis and hepatocellular carcinoma (HCC). The potential of sAXL and Gas6 as a diagnostic tool is still an open question. In multicenter studies, we evaluate the clinical utilities of sAXL and Gas6 in large cohorts of patients with chronic liver disease and HCC by comparing diagnostic accuracies with current clinically relevant methods. We aim to establish sAXL and Gas6, either on its own or integrated into biomarker panels, as effective screening and monitoring tools for patients with chronic liver disease.